- Secondary Main Malignancy charges didn’t considerably differ amongst varied CAR T-cell therapies or most cancers sorts
- Sufferers with greater than three prior traces of remedy had the next SPM danger
- CAR T-cell remedy has a comparable danger of secondary cancers to straightforward remedies, with SPM charges of 5% versus 4.9%, respectively
The U.S. Meals and Drug Administration (FDA) mandated that each one CAR T-cell therapies which are at present available on the market have a boxed warning on the label informing sufferers and prescribers about the potential for a secondary malignancy growing following CAR T-cell remedy (1✔ ✔Trusted Supply
Second major malignancies after CAR T-cell remedy: A scientific evaluate and meta-analysis of 5,517 lymphoma and myeloma sufferers
Go to supply
).
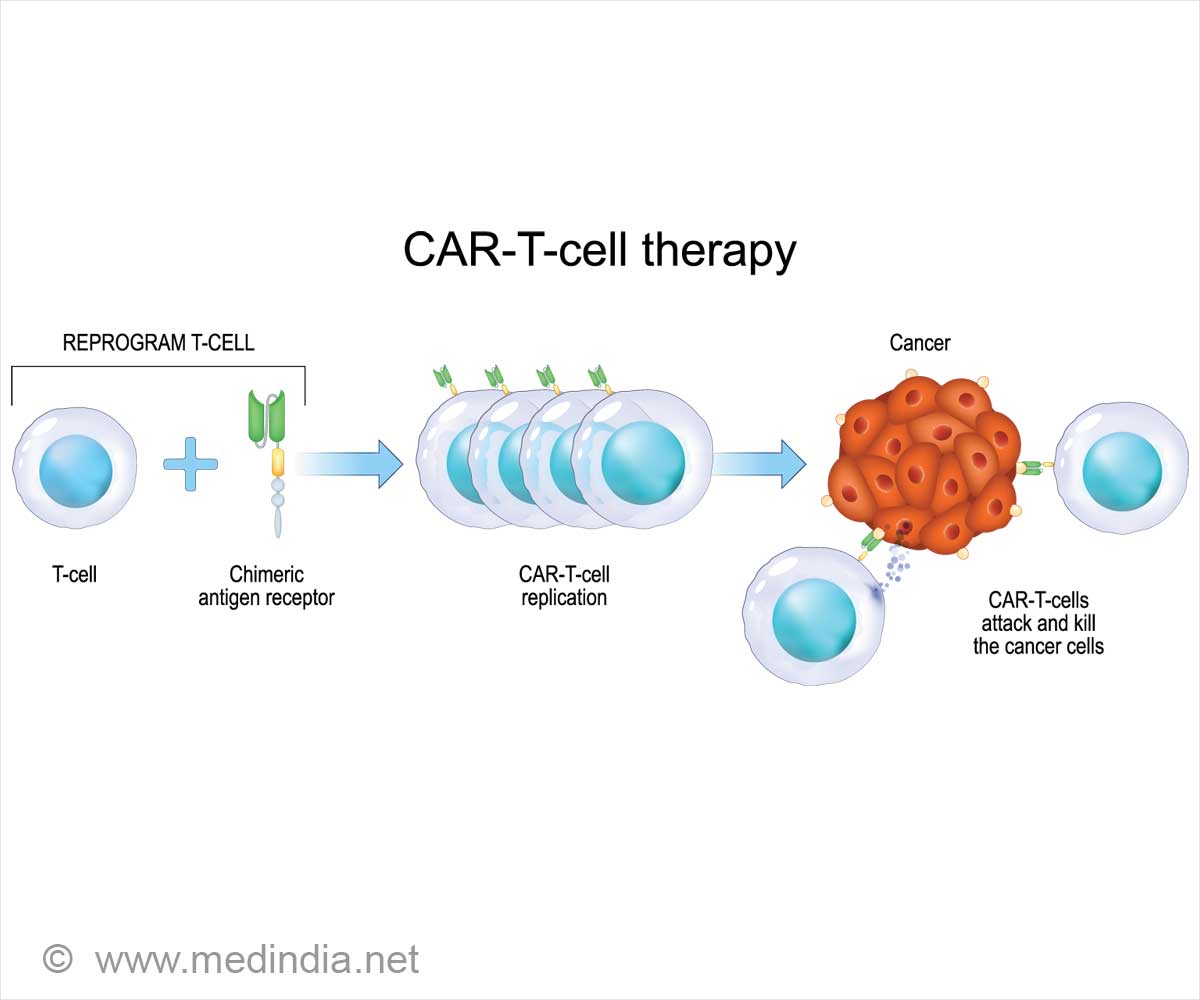
Not like chemotherapy or immunotherapy, which depend on generally produced medicine, CAR T-cell remedy makes use of a affected person’s cells. On this course of, T-cells are modified within the lab to reinforce their skill to assault tumors.
The engineered cells are infused again into the affected person’s bloodstream after being conditioned to multiply successfully. At the moment, CAR T-cell remedy is authorised for treating leukemias and lymphomas.
Sufferers receiving CAR T-cell remedy for a number of myeloma or B-cell lymphoma are particularly warned within the boxed warnings in regards to the danger of buying new T-cell malignancies unrelated to their unique circumstances.
Knowledge from the FDA Opposed Occasion Reporting System served as a significant foundation for the choice. Nevertheless, different researchers are involved that the info might include inborn flaws, resembling reporting bias.
Commercial
Key Elements in Assessing SPM Threat
When evaluating the danger of secondary major malignancies (SPMs), a number of variables had been thought of, together with:
- Age
- Size of follow-up
- Kind of CAR T-cell remedy acquired
- Preliminary analysis
- Prior remedies
Commercial
Authorised CAR T-Cell Therapies
Researchers performed a scientific evaluate and meta-analysis of scientific research to find out which of the six CAR T-cell therapies are at present authorised for grownup sufferers with lymphoma or a number of myeloma.
Examples of CAR T-cell therapies at present authorised embody:
- Tisagenlecleucel, also called tisa-cel (Kymriah)
- Axicabtagene ciloleucel, also called axi-cel (Yescarta)
- Brexucabtagene autoleucel, also called brexu-cel (Tecartus)
- Lisocabtagene maraleucel, also called liso-cel (Breyanzi)
- Idecabtagene vicleucel, also called ide-cel (Abecma)
- Ciltacabtegene autoleucel, also called cilta-cel (Carvykti)
These research included grownup sufferers who acquired the six CAR T-cell therapies.
To satisfy the inclusion standards, the research wanted to incorporate all the follow-up time, which ranged from 6.6 months to 65.4 months. A complete of 326 SPMs from 5,517 sufferers had been included within the closing choice, which comprised seven real-world investigations and 18 scientific trials.
The SPM charges didn’t considerably change throughout sufferers with totally different most cancers sorts or between sufferers who obtained totally different CAR T-cell merchandise.5.8% of sufferers skilled an SPM at a follow-up of 21.7 months.
Commercial
T-Cell Malignancies as Secondary Dangers in CAR T-Cell Remedy
Research discovered that sufferers who had undergone greater than three traces of remedy earlier than CAR T-cell had a significantly elevated danger of SPMs, in comparison with sufferers who had acquired fewer than three traces of remedy.
Equally, the speed of SPMs was 4.2% throughout research with a follow-up interval of lower than 21.7 months, and eight.5% amongst research with a follow-up time of greater than 21.7 months.
Hematologic malignancies, resembling acute myeloid leukemia and myelodysplastic syndrome, accounted for the largest share of SPMs (37%). T-cell malignancies accounted for 5 of the circumstances of the full examine inhabitants.
When analyzing the CAR T-Cell transgene in three of those circumstances, it was discovered that the malignant T cells examined constructive, suggesting that the malignancy might have originated from CAR-edited cells.
Evaluating CAR T-Cell Remedy With Customary Remedies
In 4 scientific trials, researchers in contrast CAR T-cell remedy to straightforward remedies in 1,253 sufferers. They discovered that 5% of sufferers receiving CAR T-cell remedy developed secondary cancers, in comparison with 4.9% of these on customary remedies. This small distinction wasn’t important, which means CAR T-cell remedy did not present the next danger of secondary cancers in comparison with customary care.
The examine discovered no elevated danger of secondary major malignancies (SPMs) with CAR T-cell remedy in comparison with customary remedies. The info recommend that warning labels may scare sufferers, regardless of the low danger.
Elements such because the variety of prior remedies and longer follow-up instances may affect SPM danger. CAR T-cell remedy, which has demonstrated survival advantages in treating resistant giant B-cell lymphoma, shouldn’t be prevented because of the minimal danger of recent T-cell cancers.
Additional analysis is required to grasp the position of CAR T-cell remedy in growing SPMs and to enhance particular person danger assessments. Correct long-term reporting in scientific trials is essential. The examine’s limitations embody variability within the information and lacking details about sufferers’ earlier remedies and well being circumstances, which may impression SPM danger.
Reference:
- Second major malignancies after CAR T-cell remedy: A scientific evaluate and meta-analysis of 5,517 lymphoma and myeloma sufferers – (https://aacrjournals.org/clincancerres/article-abstract/doi/10.1158/1078-0432.CCR-24-1798/747919/Second-primary-malignancies-after-CAR-T-cell)
Supply-Medindia


